This Is Your Ocean on Acid
 On most days, Bill Dewey can be found wearing waist-high waders and inspecting Manila clams—the West Coast version of the littleneck—at his Washington clam farm, Chuckanut Shellfish. Under an arrangement that’s unique to the state, Dewey owns 32 acres of tidelands. Unlike land-based farms, he can only harvest when the tide recedes, leaving over a mile of mudflats, and shellfish, exposed. He gathers the clams with the help of a former tulip-bulb harvesting machine that’s carried out aboard his boat, the Clamdango!
On most days, Bill Dewey can be found wearing waist-high waders and inspecting Manila clams—the West Coast version of the littleneck—at his Washington clam farm, Chuckanut Shellfish. Under an arrangement that’s unique to the state, Dewey owns 32 acres of tidelands. Unlike land-based farms, he can only harvest when the tide recedes, leaving over a mile of mudflats, and shellfish, exposed. He gathers the clams with the help of a former tulip-bulb harvesting machine that’s carried out aboard his boat, the Clamdango!
Working on the mudflats, often with his son and dog in tow, is the fulfillment of a dream for Dewey, a shellfish farmer for more than 30 years who is also the public policy and communications director for Taylor Shellfish Company. Taylor’s operations—which include growing oysters, clams, mussels and geoduck (giant clams whose necks can reach more than three feet long)—span some 1,900 acres of the same tidelands. All told, there are about 47,000 acres of oceanic land that have that special designation in the state, and, he says, “It’s fundamental as to why Washington leads the country in farmed shellfish production. In other parts of country, you typically have to lease the land from the state. Banks are less apt to loan money to businesses that have to lease.”
Commercial shellfishing makes up the lion’s share—two-thirds—of the nation’s aquaculture industry. So reports the National Oceanic and Atmospheric Administration’s (NOAA’s) Fisheries Service which makes a case for boosting domestic seafood production, noting that Americans eat a lot of seafood, and import 86% of it, creating a U.S. seafood trade deficit that now exceeds $10.4 billion annually, second only to oil when it comes to natural resources. In the Pacific Northwest, the shellfish industry contributes $270 million per year to the regional economy and employs more than 3,200 people. And when oyster cultivation fails at the top Northwest hatcheries and farms, the effects on the industry are devastating.
A SHELLFISH STORY
 For centuries, shellfish farmers have cultivated oysters in Washington’s Willapa Bay, a massive, shallow estuary separated from the Pacific Ocean by the Long Beach Peninsula. The bay’s warm waters are particularly suited for growing Pacific oysters, identified by their rough, fluted shells marked with purple streaks, and a white interior bearing “a single muscle scar that is sometimes dark, but never purple or black,” according to a Biological Report from the U.S. Fish and Wildlife Service. The oyster was imported from Japan to the western U.S. coast in 1903. “Puget Sound and Washington waters are a little bit cold compared to what the oyster had in Japan,” says Dewey. “So it doesn’t reproduce particularly well here. Except for a few areas—Willapa Bay is one of them. There’s dependable natural reproduction from one year to the next. The water basically has to get up to 72 degrees and stay there for three weeks for the oysters to spawn.”
For centuries, shellfish farmers have cultivated oysters in Washington’s Willapa Bay, a massive, shallow estuary separated from the Pacific Ocean by the Long Beach Peninsula. The bay’s warm waters are particularly suited for growing Pacific oysters, identified by their rough, fluted shells marked with purple streaks, and a white interior bearing “a single muscle scar that is sometimes dark, but never purple or black,” according to a Biological Report from the U.S. Fish and Wildlife Service. The oyster was imported from Japan to the western U.S. coast in 1903. “Puget Sound and Washington waters are a little bit cold compared to what the oyster had in Japan,” says Dewey. “So it doesn’t reproduce particularly well here. Except for a few areas—Willapa Bay is one of them. There’s dependable natural reproduction from one year to the next. The water basically has to get up to 72 degrees and stay there for three weeks for the oysters to spawn.”
Beginning in 2005, these oysters in the bay, known as natural sets, stopped reproducing. They have never successfully reproduced since. In 2006, the hatchery-produced Pacific oysters followed suit. In the hatcheries, spawning happens year-round in conditioning tanks where water temperature and algae levels (for food) are closely controlled.
Both Taylor Shellfish and Whiskey Creek Shellfish Hatchery in Tillamook, Oregon, witnessed oyster larvae die-offs that they couldn’t explain and that continued for years. Initially, they suspected a bacteria known as Vibrio tubiashii was to blame. But even after Whiskey Creek installed an expensive filtration system, the oyster larvae continued to die. By 2008, Whiskey Creek, which alone accounts for 75% of all oyster seedlings used by West Coast oyster farmers, had lost 80% of its oyster larvae. Taylor Shellfish had lost 60%. Despite the controlled environment, the ocean water they were pumping into their hatcheries was corrosive. Upwelling—or deep ocean water rising to the surface following north winds off the Washington coast—was carrying acidic water to the surface. The shellfish farmers were experiencing the devastating impacts of ocean acidification sooner than researchers had anticipated. With support from Senator Maria Cantwell (D-WA), ocean acidification sensors were set up in 2010 near Washington’s hatcheries. Combined with Integrated Ocean Observing System (IOOS) buoys from NOAA measuring wind velocity, they track ocean acidity—and predict the upwelling events that cause increased acidity—in real time.
Mark Wiegardt, co-owner of Whiskey Creek said: “Putting an IOOS buoy in the water is like putting headlights on a car.” Adds Dewey: “All of a sudden we could see all aspects of this water that was coming in our intake pipes. And it was quite eye-opening. We were seeing pH levels down as low as 7.5. Normally it’s 8.2.” To oyster larvae, it’s the difference between life and death.

When that acidic water entered the hatcheries, it caused oyster shells in their critical formative period to dissolve. Oysters and other shellfish, including clams and lobsters, and a host of sea creatures that include plankton and corals, need calcium carbonate minerals to form their shells and skeletons. Normally ocean water is full of these minerals, but as carbon dioxide (CO2) emissions have climbed across the globe, the ocean has absorbed increasing levels of CO2, causing ocean acidification to rise and the availability of these minerals to fall
“A lot of things we like to eat have these calcium carbonate shells and they’re very sensitive to acidification,” says Richard Feely, Ph.D., a senior scientist with NOAA and its Pacific Marine Environmental Laboratory (PMEL). “Just a small drop in pH can cause the shells to begin to dissolve. It turns out that for many of these species, the larval and juvenile stages are much more sensitive than the adults. And we’re finding that they can die off quite rapidly even with the kinds of changes that we’re seeing right now.”
SWALLOWING EMISSIONS
Over the past 100 years, levels of carbon in the atmosphere have risen 30%—to 393 parts per million. And the oceans absorb a third of that carbon dioxide, or approximately 22 million tons per day, in a process that Feely likens to adding carbon to water to make soda. Once it sinks into the water, the carbon dioxide reacts with water molecules to form carbonic acid; the carbonic acid then releases hydrogen ions which in turn combine with carbonate ions (the ones that shellfish and other creatures need) removing them from the water. Normally the process of oceans soaking up our excess CO2 is a beneficial one—keeping global warming in check. “Eventually, over a very long time, thousands of years, the ocean will take up 85-90% of all the carbon that’s released,” says Feely. “We thought that was a good thing.” But acidification is now happening at an accelerated pace, and it’s already changing the ocean in profound ways.

A study published in Science in March 2012 found that ocean acidity may be increasing faster today than it has during four major extinctions in the last 300 million years. The only time period that remotely resembles the ocean changes happening today, based on geologic records, was 56 million years ago when carbon mysteriously doubled in the atmosphere, global temperatures rose by approximately six degrees and ocean pH dropped sharply, driving up ocean acidity and causing a mass extinction among single-celled ocean organisms. It’s likely, researchers surmise, that higher organisms also disappeared as a result. During that extinction period, ocean pH levels fell by up to 4.5 units. In the last 100 years, ocean pH today has already fallen by .1 unit—10 times faster than during that extinction period—and could drop another .3 units by the end of the century if predictions from the Intergovernmental Panel on Climate Change are correct. Such a drop in pH, says Feely, “would increase the acidity of the ocean by about 100% to 150%. That’s a dramatic change.”
The oyster die-offs are likely just the first sign of significant impacts to come if carbon emissions aren’t reined in. Take, for example, the pteropod or sea butterfly. These tiny marine snails that appear winged and beautifully translucent in close-ups are essential to the ocean food web. Ocean acidification threatens the ability of pteropods to form their fragile shells, putting a range of commercially important fish at risk that depend on the small snails for food, including salmon, herring and yellowfin tuna as well as mammals like baleen whales, ringed seals and marine birds. Scientist Gretchen Hofmann of the University of California Santa Barbara said of pteropods to United Press International: “These animals are not charismatic, but they are talking to us just as much as penguins or polar bears. They are harbingers of change. It’s possible by 2050 they may not be able to make a shell anymore. If we lose these organisms, the impact on the food chain will be catastrophic.”
CORAL COLLAPSE
Corals, too, face direct threat from ocean acidification, which, as it robs ocean water of carbonate ions, impedes their ability to form skeletons. Davey Kline, Ph.D., a coral reef ecology expert at the University of Queensland in Australia, first began diving in the Caribbean in 1997 and says at that time, “there were still really beautiful, elaborate reefs with really high coral coverage. Corals bigger than me that looked like giant trees forming a forest. But in the 10 years I’ve been working in the Caribbean, I saw those once really incredible reefs completely crash and disappear. And what were once these really diverse, three-dimensional reef structures became seaweed beds. Where the corals were gone, most of the fish were gone and all that was left was a lot of stinging, nasty algae.”

It’s not just ocean acidification threatening these reefs, it’s a number of factors including overfishing, disease, development and warming waters. But the falling pH has a very specific impact on the corals’ ability to grow, making it that much more difficult for them to withstand other stressors. Kline describes the growth and erosion of coral reefs as “a really delicate balance.” Corals are built by polyps—tiny anemone-like creatures that produce calcium carbonate crystals, stacking them in intricate, interconnected branches faster than the sea can erode their skeletons. “There have been a lot of studies showing that under ocean acidification scenarios that corals and other organisms on the reef calcify at a slower rate,” Kline says. “Even with just a little less growth, the corals can be tipped into these situations where they’re getting eroded faster than they can grow and the reefs start to dissolve.”
It is nearly impossible to quantify the importance of coral reefs to people and the planet. In monetary figures, corals have been valued at $29.8 billion per year in net global economic value because they support fisheries, tourism and all the associated businesses, from hotels to restaurants. Reefs also protect shorelines from damaging storm waters and prevent erosion; they are the rainforests of the sea that provide a home for one million species; and they are “the medicine cabinets of the 21st century” according to NOAA’s Coral Reef Conservation Program, providing new sources of medicine to treat cancer, HIV, heart disease, arthritis and other diseases. Reefs are thriving underwater metropolises where fish spawn and hide from predators and bigger fish cruise looking for food.
Sponges, the most primitive reef animals, house tiny fish in their cavernous tubes and vases as they draw seawater into their pores. The critically endangered hawksbill turtle, with its almond eyes, black spots and hooked beak, rests on the reefs feeding on these sponges while the vulnerable dugong, a flabby mammal with a wide snout and dolphin-esque tail, circles lagoons, feeding on the reef’s seagrasses. Shrimp and crabs are ubiquitous in coral reef environments around the world, hiding in crevices, providing cleaning services and enjoying the ready food supply. And of course the fish, of every hue and size and shape, with bodies designed to quickly maneuver through reef structures, fend off predators with scalpel-like spines, scrape algae and avoid stinging tentacles, all coexist in these incredible habitats.
“If we lose coral reefs we lose a substantial source of seafood for coastal countries in the tropics in particular,” says Mark Spalding, president of the Ocean Foundation. “You’re threatening the basic productivity of the ocean.”
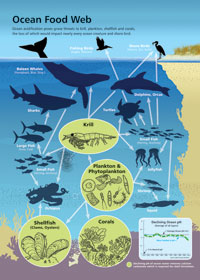
CLICK HERE to view full-size version of Ocean Food Web info graphic.
And the potential for a world without coral reefs is not far-fetched or far off. The most recent report on reef health—Status of Coral Reefs of the World: 2008—found that 19% of coral reefs were already lost, 15% were seriously threatened within a decade or two, and 20% could be lost in 20 to 40 years. “If we continue on the trajectory that we’re currently at,” says Kline, referring to unchecked global emissions, “we will lose reefs as we know them. We’ll probably see a transition from really diverse reefs to reefs with fewer species that are tougher, weedier species that can deal with these dramatic conditions. Associated with the loss of diversity of corals will be the loss of millions of species that use corals as their homes. A lot of the fish and seafood that we eat, the most critical part of their life stages are on coral reefs. So there will be huge economic impacts in terms of loss of fisheries, loss of sustenance for all the cultural communities and loss of tourism…These changes could all happen within the next 30 or 40 years—by 2050, at the current rate of change.”
Increased carbon dioxide in the atmosphere not only alters the ocean’s chemistry, it’s increasing the temperature of the atmosphere and warming waters, too. As ocean temperatures rise, a very important algae called zooxanthellae (zoo-zan-thel-y) that provides food for corals—and contributes to their remarkable colors—can no longer make food. That’s when corals bleach. “The reason the corals become bright white is because most of their color is coming from these algae,” says Kline. “And when they lose the algae because the water is too warm and they can’t keep up this relationship anymore, you see the bare skeleton.”
Sometimes bleaching happens en masse as when 95% of corals in the Philippines bleached in 2010 after an El Niño event that raised ocean temperatures. Increased ocean temperatures also make the waters more stratified—preventing nutrient-rich water from below from rising to the surface and oxygen-rich water from reaching the middle layers. This can lead to more widespread losses. The Center for Ocean Solutions writes: “Between 1951 and 1993 zooplankton biomass off Southern California decreased by 80% as a result of warming surface waters.” Less oxygen reaching the interior, meanwhile, a product of both this increased stratification and significant nutrient runoff from farms, creates dead zones, a massive threat to marine life. And unlike nutrient runoff, which can be brought under control rather quickly, oxygen depletion that happens as a result of global warming can’t be easily reversed.
“Ocean warming, acidification and deoxygenation are essentially irreversible on centennial time scales,” found the Royal Society, a London-based group specializing in scientific research, in a 2011 paper, “[O]nce these changes have occurred, it will take centuries for the ocean to recover. With the emission of CO2 being the primary driver behind all three stressors, the primary mitigation strategy is to reduce these emissions.”

CLICK HERE to view full-size version of The Great Barrier Reef info graphic.
PIECEMEAL SOLUTIONS
It would be hard to find an ocean expert who does not agree that global carbon dioxide emissions must be brought under control—and quickly—if we are to prevent the wholesale deterioration of our oceans. Most also recognize that such global agreements are the most difficult to come by, and that local protection strategies and efforts to reduce stressors on corals and marine life are important steps in at least staving off the impacts of ocean acidification and global warming.
When it comes to reefs, designating reef environments as marine protected areas (MPAs)—and enforcing that designation—is essential to protecting habitat. But, notes the World Resources Institute, of the 400 or more MPAs in more than 65 countries and territories, there are only a handful that are truly large in scope—notably the Great Barrier Reef in Australia, the Florida Keys National Marine Sanctuary and the Ras Mohammed Park Complex in Egypt. Outside of these massive sites, they write “it is likely that less than 3% of the world’s coral reefs are protected.” And in many cases, such protections are on paper only. They cite the example of Johnston Atoll west of Hawaii, which was designated a federal bird refuge in 1926, and became the Pacific Marine National Monument under President George W. Bush in 2009. “Probably among the earliest designations of a coral reef protected area, this site has been subjected to massive military development, high atmospheric nuclear testing, chemical waste disposal, and other threats,” the institute notes.
At the Great Barrier Reef, the world’s largest coral reef ecosystem at approximately 133,000 square miles (about the size of New Zealand), establishing the Great Barrier Reef Marine Park in 1975 was a first step, but not until the park was rezoned between 1999 and 2003 was the reef given the protection needed to rebound from threats that include shipping, dredging, commercial fishing, nutrient and pesticide runoff, coastal development and diving. Some 33% of the Great Barrier Reef is designated as a Green Zone, or no-take zone, where any activity beyond diving and underwater photography is prohibited or requires a permit. There are seven zones in total, which allow varying degrees of fishing, aquaculture, trawling and other activities, keeping them within managed limits. The restrictions have led to major recoveries of reef fish—including the coral trout and stripy sea perch—and declines in the crown-of-thorns starfish, a large starfish with up to 21 arms that lives and preys on corals, killing them in the process. The crown-of-thorns starfish proliferates in nutrient-rich water which comes as a result of unchecked runoff.
“As much of the stress as you can remove from reefs you’re really going to increase the chance that more of the reefs can make it,” says Kline. “Setting up marine reserves and managing marine reserves well; minimizing pollution and development near reefs; and using reefs in a sustainable way. Corals are living animals and when people step on them or kick them with their fins it can cause damage to the reefs. All these different factors can have an impact on the overall future of coral reefs.”
Shellfish farmers with controlled hatchery environments can take some precautions to prevent corrosive, acidic water from entering their breeding tanks. Thanks to ocean buoys and sensors monitoring acidity and wind velocity, farmers at Whiskey Creek now know that they have 24 hours following a north wind before corrosive water wells up and enters their intake pipes. “When they see [a north wind] happening,” says Dewey, “they fill all their tanks and they don’t change their water as frequently as they should to avoid bringing corrosive water in that would harm the larvae. They’ve adapted management protocols to get around those corrosive events that are somewhat effective.”
But in order to track and manage ocean acidification more monitoring is needed, and the federal 2013 budget cuts $2.5 million in funding for obtaining and delivering data from the buoys in Washington state. That led Sen. Cantwell—who sits on the Energy and Natural Resources Committee—to confront NOAA Administrator Jane Lubchenco at a March 7, 2012 hearing, saying: “Cutting back on science that is important for jobs and the economy can’t be substituted.” Lubchenco admitted during the hearing that cutting the funding for ocean acidification monitoring “is one of those choices that I’m not happy about because it’s a program that is very, very important. We will continue to do monitoring; it’s not that we’re not doing anything. We won’t be able to do it at the scale we would like to do it.”

Other fixes shellfish hatcheries can employ include filling the tanks later in the day, when the water has warmed and the pH has increased, and running the water over clam or oyster shells before filling tanks, which also increases pH. It’s an imperfect process, but workable, for now.
What is critical to reducing the effects of ocean acidification surrounding coasts, says Spalding, is to protect and restore seagrass. Florida’s coasts, for example, have lost significant seagrass, in large part from dredge and fill operations. This seagrass is not only essential to provide habitat for fish, but the plants store CO2 in their roots, lowering the ocean’s pH. Mangroves, which are “forested wetlands,” serve the same function, and are similarly threatened, particularly by shrimp aquaculture. Since the 1980s, 20% of the world’s mangroves have been destroyed, according to the Food and Agriculture Organization of the U.N.
“One solution [to ocean acidification] is to make sure that we do everything we can to preserve and protect salt marshes, sea grasses and mangroves in particular,” Spalding says, “and be aggressive about restoring those that we’ve lost to recreate the carbon sink potential of the ocean.” If this restoration happened on a global level, it could help lower the pH overall; and there’s speculation, Spalding adds, that such strategies might work to control the pH of individual areas.
As the Royal Society noted, however, the only real, overarching solution to ocean acidification is setting significant global targets for reducing CO2 emissions and sticking to them. In lieu of that, it means local communities—particularly coastal “hotspots”—must adopt ways to address ocean acidification using existing laws, according to a May 2011 report by Feely and other experts. That includes enforcing the federal Clean Water Act which requires the control of pollutants and runoff (both of which increase acidification), enacting zoning policies that address runoff and emissions and enforcing federal laws on emission limits.
These local strategies, Feely says, may offer the only immediate possibility for mitigating ocean acidification. In terms of setting reduced targets for worldwide carbon dioxide emissions, he says, we’ll almost certainly pass the “safe” point from the oceans’ perspective. “One of the problems we’re faced with is trying to figure out what’s a safe level for CO2,” Feely says. “And many folks have suggested that we would like to keep global warming below a level of total increased temperature of 2°C. To do that, you have to have CO2 levels in the atmosphere below 450-500 parts per million. A CO2 concentration of 450-500 ppm means the Arctic Ocean and good portions of the Antarctic Ocean would become corrosive to all calcifying organisms from surface to bottom. In fact, from the retrospect of ocean acidification, we’ll reach thresholds long before we get to those levels.”



